How Many Valence Electrons In Oxygen? A Complete Guide To Electron Configuration And Bonding
Have you ever wondered how many valence electrons in oxygen are responsible for its incredible versatility in nature? From the air we breathe to the water that sustains life, oxygen's chemical behavior is dictated by a simple yet profound number: its valence electrons. Understanding this concept isn't just academic—it's the key to predicting how elements interact, form molecules, and create the building blocks of our world. Whether you're a student tackling chemistry for the first time or a curious learner, this guide will demystify oxygen's valence electrons, explore its electron configuration, and reveal why this knowledge is fundamental to chemistry.
We'll start with the direct answer: oxygen has 6 valence electrons. But that's just the beginning. This number explains why oxygen is so reactive, why it forms two bonds in most compounds, and why water has its unique bent shape. By the end of this article, you'll not only know the answer but understand the "why" behind it, empowering you to determine valence electrons for any element and grasp the elegant logic of the periodic table.
What Are Valence Electrons? The Outer Shell's Secret Power
Before diving into oxygen, let's define our star player. The outermost electrons in each atom are called valence electrons. These are the electrons in the highest energy level (or shell) of an atom, and they are the sole participants in chemical bonding. Think of them as an atom's "social" electrons—they're the ones that interact, share, donate, or accept when atoms come together to form molecules. The number of valence electrons an atom has is determined primarily by its position on the periodic table, specifically its group number for main group elements.
Why does this matter? The number of valence electrons determines how an atom interacts with others, including how many electrons it needs to gain or lose to achieve a noble gas configuration. This drive for stability—often an octet (eight electrons) in the outer shell, akin to noble gases like neon or argon—is the engine of chemical reactions. For oxygen, with its six valence electrons, this means it's "two electrons short" of a stable octet, shaping its entire chemical identity.
Finding Valence Electrons: Two Reliable Methods
You don't need to memorize every element. There are two straightforward ways to find the valence electrons of any element in the periodic table.
Method 1: The Group Number Shortcut
For main group elements (groups 1, 2, and 13-18), the group number often indicates the number of valence electrons.
- Group 1 (IA): 1 valence electron (e.g., Sodium, Na)
- Group 2 (IIA): 2 valence electrons (e.g., Magnesium, Mg)
- Group 13 (IIIA): 3 valence electrons (e.g., Aluminum, Al)
- Group 14 (IVA): 4 valence electrons (e.g., Carbon, C)
- Group 15 (VA): 5 valence electrons (e.g., Nitrogen, N)
- Group 16 (VIA): 6 valence electrons (e.g., Oxygen, O)
- Group 17 (VIIA): 7 valence electrons (e.g., Fluorine, F)
- Group 18 (VIIIA): 8 valence electrons (e.g., Neon, Ne) – these are the noble gases, already stable.
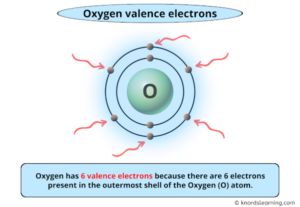
Oxygen is in group 16 (also known as group 6A) of the periodic table, which means it has 6 electrons in its outermost energy level. This is the fastest way to answer our key question.
Method 2: Electron Configuration
For a more fundamental understanding, you can write the atom's electron configuration. This shows how electrons are distributed in atomic orbitals. The electrons in the highest principal energy level (the highest "n" number) are the valence electrons.
Looking at the electron configuration, oxygen has 6 electrons in its outermost shell (the second shell, with the 2s and 2p orbitals). The full configuration for oxygen (atomic number 8) is 1s² 2s² 2p⁴. The first shell (n=1) holds 2 electrons (1s²), but these are core electrons, not involved in bonding. The second shell (n=2) holds the remaining 6 electrons (2s² 2p⁴). Therefore, these 6 electrons are the valence electrons.
Pro Tip: You can use an interactive chart to visualize this. Many online tools let you click an element and see its electron configuration highlighted, showing valence vs. core electrons instantly.
Oxygen's Valence Electrons: A Deep Dive
Now let's focus specifically on oxygen, combining the methods above.
Oxygen's Position and Classification
Oxygen has 6 valence electrons. It sits proudly in Group 16, the chalcogens. Oxygen is classified as a nonmetal, more specifically, a reactive nonmetal. Its high electronegativity and desire to gain electrons stem directly from having 6 valence electrons—just two shy of a stable octet. This makes oxygen one of the most electronegative elements (second only to fluorine), meaning it has a strong pull on shared electrons in bonds.
Oxygen has 8 electrons in total, but only 6 of them are valence electrons. The other 2 are in the innermost 1s orbital, tightly bound to the nucleus and not involved in chemistry. This distinction is crucial: total electrons include all electrons, while valence electrons are only the outermost ones.
Visualizing Oxygen's Valence Electrons: Lewis Dot Structure
Chemists use Lewis dot diagrams to represent valence electrons as dots around the element symbol. For oxygen, with 6 valence electrons, the Lewis symbol is:
.. O .. (Or more commonly written as Ö or O with two dots on each side). Lewis dot diagrams are primarily used to represent which type of? They are used to represent the valence electrons of an atom, primarily for predicting covalent bonding and molecular structure.
The Octet Rule: Oxygen's Quest for Stability
With six valence electrons, oxygen needs to gain or share two additional electrons to complete its octet. This is where the famous octet rule comes in. The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons (or two for hydrogen and helium), resembling a noble gas configuration.
The octet rule is a guiding principle, but it is not universally applicable. While it works perfectly for oxygen and many main group elements, there are exceptions (like boron with 6 electrons or elements in period 3+ that can have expanded octets). However, for oxygen, the rule is almost always followed.
How Oxygen Achieves an Octet: Bonding Strategies
Oxygen commonly achieves this stability by forming two chemical bonds. There are two primary ways:
Covalent Bonding (Sharing): Oxygen shares one electron with two other atoms, forming two single bonds. In a water molecule (H₂O), oxygen shares one electron with each hydrogen atom, giving oxygen a share in 4 electrons from the bonds plus its own 4 non-bonding electrons, totaling 8. With six valence electrons, oxygen needs to gain or share two additional electrons to complete its octet—sharing does this efficiently.
Ionic Bonding (Gaining): Oxygen can gain two electrons to become an oxide ion (O²⁻). This would add 2 electrons to its normal configuration making the new configuration 1s² 2s² 2p⁶, which is exactly the same as neon's. With 10 electrons you should note that oxygen's electron configuration is now exactly the same as neon's, achieving a stable noble gas configuration. This is common in metal oxides like MgO.
Comparing Oxygen to Other Elements
Understanding oxygen's 6 valence electrons is clearer when compared to its neighbors.
| Element | Symbol | Group | Valence Electrons | Common Bonds |
|---|---|---|---|---|
| Hydrogen | H | 1 | 1 | 1 |
| Carbon | C | 14 | 4 | 4 |
| Nitrogen | N | 15 | 5 | 3 (or 4 with charge) |
| Oxygen | O | 16 | 6 | 2 |
| Fluorine | F | 17 | 7 | 1 |
Solution for how many valence electrons are represented in the Lewis dot symbol for a nitrogen atom? Nitrogen is in group 15, so it has 5 valence electrons. The answer is c) 5.
How many bonds does carbon want to make with other atoms? Carbon has 4 valence electrons and needs 4 more for an octet, so it typically forms 4 bonds (as in methane, CH₄).
How many valence electrons does carbon have? 4. This is why organic molecules must have which atoms? The defining feature is carbon and hydrogen, though oxygen, nitrogen, etc., are also common.
If you knew the number of valence electrons in a nonmetal atom, how would you determine the valence of the element? For main group nonmetals, the typical number of bonds equals (8 - number of valence electrons). For oxygen: 8 - 6 = 2 bonds. For nitrogen: 8 - 5 = 3 bonds.
Molecular Geometry: The Water Molecule Case Study
Oxygen's valence electrons not only dictate how many bonds it forms but also the shape of the molecules it creates. Let's examine water (H₂O).
What is the electronic geometry structure of the water molecule? Oxygen has 6 valence electrons. It uses 2 to form bonds with two hydrogen atoms. The remaining 4 electrons exist as two lone pairs. This gives four regions of electron density (2 bonding pairs + 2 lone pairs). The electronic geometry is tetrahedral.
How many pairs of valence electrons are there on the oxygen atom in the water molecule? There are 4 pairs total: 2 bonding pairs and 2 lone pairs.
What is the approximate H—O—H bond angle in water? The ideal tetrahedral angle is 109.5°, but lone pairs repel more strongly than bonding pairs. What is the actual bond angle? The H—O—H bond angle is approximately 104.5°, a direct result of oxygen's two lone pairs pushing the hydrogen atoms closer together.
This bent shape is crucial for water's unique properties, like its high boiling point and ability to dissolve many substances—all stemming from oxygen's 6 valence electrons and the resulting lone pairs.
Beyond Oxygen: Interactive Learning and Common Questions
Using Interactive Tools
The mention of "Drag an electron from the left atom to the right atom" and "Click pause, drag an electron from the right atom to the left, and then click play" refers to digital simulations that model electron transfer. These are fantastic for visualizing ionic bonding (e.g., sodium giving an electron to chlorine) or covalent bonding (sharing). For oxygen, you could simulate it gaining two electrons from two sodium atoms to form Na₂O, or sharing electrons with hydrogens.
Reproduction for educational use only—these tools are widely available on sites like PhET Interactive Simulations and are perfect for classroom or self-study.
Addressing Common Questions
How many valence electrons does O²⁻ have? The oxide ion has gained 2 electrons, so it now has 8 valence electrons (same as neon). It has a full octet.
How many valence electrons are in an oxygen molecule (O₂)? Each oxygen atom in O₂ still has 6 valence electrons. However, in the covalent bond, they share 4 electrons (a double bond), so each oxygen "counts" 8 electrons around it (4 from the bond + 4 unshared), satisfying the octet rule.
Why doesn't oxygen form three bonds like nitrogen? Nitrogen has 5 valence electrons and needs 3 more, so it forms 3 bonds (with a lone pair). Oxygen has 6 and needs 2 more, so it forms 2 bonds (with two lone pairs). Forming three bonds would give oxygen a formal charge of +1, which is less stable.
The Bigger Picture: From Valence Electrons to Molar Mass
While valence electrons govern bonding, other concepts like molar mass are based on atomic mass. How many grams of carbon equal one mole of carbon? Carbon's atomic mass is approximately 12.01 amu, so approximately 12 grams of carbon is equal to 1 mole (more precisely, 12.01 g). How many grams of NaCl equal a mole of NaCl? Sodium (23) + Chlorine (35.45) = 58.45 g/mol.
The number of moles in a substance is determined using the substance's molar mass, the number of grams in one mole of a substance. This connects back to atoms: one mole of oxygen atoms contains 6.022 x 10²³ atoms, each with 8 protons/neutrons (roughly 16 g/mol for atomic oxygen) and, of course, 6 valence electrons per atom. So while valence electrons don't affect molar mass, they define how those atoms combine—e.g., one mole of O₂ molecules (32 g) contains two moles of oxygen atoms, each with 6 valence electrons participating in a double bond.
Conclusion: The Foundational Importance of Valence Electrons
So, how many valence electrons in oxygen? The definitive answer is 6. This single number, derived from its position in group 16 of the periodic table or its electron configuration (1s² 2s² 2p⁴), explains nearly everything about oxygen's chemistry:
- It is a reactive nonmetal with high electronegativity.
- It needs two electrons to complete its octet.
- It commonly forms two chemical bonds (as in H₂O) or gains two electrons to form O²⁻.
- In molecules like water, its two lone pairs create a bent geometry with a ~104.5° bond angle.
- Its Lewis dot structure shows 6 valence electrons, guiding predictions of molecular structure.
Mastering valence electrons unlocks the periodic table's logic. You can now look at any main group element, find its group, and immediately know its valence electrons—and by extension, its typical bonding behavior. This knowledge is the foundation for understanding chemical reactions, molecular geometry, and the composition of everything from the air we breathe to the medicines we take. The valence electrons of any element are not just a number; they are the story of that element's chemical soul, written in the language of the periodic table. Use the interactive charts, practice with Lewis structures, and soon, you'll see the elegant pattern that connects all of chemistry.